For decades, d-α-tocopheryl polyethylene glycol 1000 succinate (TPGS), a derivative of vitamin E, has been a cornerstone in oral drug formulations, prized for its multifaceted capabilities as a solubilizer, emulsifier, absorption enhancer, and bioavailability promoter, particularly for challenging, poorly soluble compounds. However, recent advancements in parenteral formulation science are revealing the profound potential of TPGS beyond oral administration. Its amphiphilic structure, ability to inhibit efflux pumps, membrane-modulating effects, and capacity to stabilize pharmaceutical actives are now expanding its utility into complex injectable modalities, including cutting-edge oncology therapeutics and the rapidly developing field of mRNA delivery platforms. This evolution signifies a paradigm shift, positioning TPGS as a critical excipient for the next generation of parenteral medicines.
A Legacy in Oral Delivery, A Future in Injectables
The journey of TPGS began over sixty years ago, establishing itself as a safe and highly effective excipient. Its inherent amphiphilic nature, characterized by a lipophilic tocopherol core and a hydrophilic polyethylene glycol (PEG) succinate tail, allows it to interact favorably with both hydrophobic active pharmaceutical ingredients (APIs) and aqueous environments. This dual characteristic has been instrumental in improving the dissolution rates, stability, and ultimately, the systemic absorption of numerous poorly soluble drugs taken orally. Commercial products like VIEKIRA PAK®, fenofibrate capsules, and ZEPATIER® feature TPGS, underscoring its proven efficacy and safety profile in established oral drug delivery systems. Its frequent use in early-stage research further highlights its value in overcoming formulation hurdles.
The transition of TPGS into the parenteral realm represents a significant expansion of its application. In injectable formulations, its hydrophobic tocopherol moiety proves adept at encapsulating lipophilic drugs, while the PEG-succinate segment ensures aqueous dispersibility and provides crucial steric stabilization. This dual functionality is particularly valuable for drugs that are difficult to formulate into stable, injectable solutions or suspensions. Furthermore, TPGS’s ability to inhibit P-glycoprotein (P-gp), a key factor in multidrug resistance (MDR), and to modulate cell membrane permeability, makes it an attractive component for advanced drug delivery systems, especially in oncology and for novel therapeutic modalities like nucleic acid-based medicines.
Pioneering Parenteral Applications: From Emulsions to Nanoparticles
The application of TPGS in parenteral products is not entirely novel, with several marketed and investigational products demonstrating its efficacy. A notable example is Solfredoc® (Docetaxel Injectable Emulsion), developed by Sayre Therapeutics. This ready-to-use docetaxel emulsion (20mg/mL) was specifically engineered to eliminate polysorbate 80 and ethanol, two excipients often associated with hypersensitivity reactions and toxicity. In Solfredoc®, TPGS serves as the primary emulsifier, facilitating an ethanol-free oil-in-water system that enhances patient tolerability and simplifies administration. Currently marketed in India for various cancers, Solfredoc® exemplifies the successful substitution of TPGS for traditional, more problematic excipients in approved oncology products, paving the way for wider adoption.
Another significant, albeit ultimately unsuccessful, endeavor was TOCOSOL® Paclitaxel Nano-emulsion by Sonus Pharmaceuticals. This platform aimed to develop a Cremophor EL®-free and ethanol-free paclitaxel formulation. The nano-emulsion incorporated α-tocopherol, TPGS, and Poloxamer 407, resulting in approximately 100-nm droplets with a high drug loading of 10mg/mL. While the Phase III clinical studies for TOCOSOL® did not meet their primary endpoints, the early-stage research provided crucial validation for TPGS as a robust emulsifier capable of stabilizing potent cytotoxic agents for parenteral administration, laying critical groundwork for future developments.
Expanding Horizons: Nanoparticle Delivery Systems
The versatility of TPGS extends significantly into the domain of nanoparticle-based parenteral formulations. Its well-established role in stabilizing conventional nanoparticles for small-molecule and anticancer therapeutics is now being extrapolated to emerging nucleic acid delivery systems. In polymeric nanoparticles, liposomes, nanocapsules, and solid lipid nanoparticles, TPGS contributes to enhanced colloidal stability, improved drug loading, and modulated pharmacokinetic profiles. These properties are achieved through its amphiphilic structure and the steric hindrance provided by its PEG chain, which can also reduce opsonization and clearance by the reticuloendothelial system (RES).
The fundamental principles governing the performance of conventional nanocarriers—protecting the therapeutic payload from degradation, preventing aggregation, and optimizing biodistribution—are directly transferable to nucleic acid-based therapeutics like messenger RNA (mRNA) and small interfering RNA (siRNA). This has spurred considerable interest in applying TPGS beyond its established uses, particularly in the context of these next-generation therapies where stability and efficient intracellular delivery are paramount challenges.
TPGS in Conventional Nanoparticle-Based Drug Delivery Systems
TPGS is a frequently employed component in various nanoparticle-based parenteral formulations, including polymeric nanoparticles, lipid nanoparticles (LNPs), and solid lipid nanoparticles. Its inclusion offers several key advantages:
- Enhanced Drug Encapsulation and Loading: The amphiphilic nature of TPGS allows it to interact with both hydrophobic drugs and the nanoparticle matrix, facilitating higher drug loading efficiencies.
- Improved Colloidal Stability: The PEG component of TPGS provides steric repulsion between nanoparticles, preventing aggregation and maintaining particle size distribution, which is critical for intravenous administration.
- Modulated Pharmacokinetics: TPGS can influence the release kinetics of encapsulated drugs and potentially prolong circulation time by reducing interactions with the immune system.
- Efflux Pump Inhibition: For cytotoxic drugs, TPGS’s ability to inhibit P-gp can help overcome multidrug resistance, thereby enhancing therapeutic efficacy.
Representative studies have demonstrated these benefits. For instance, TPGS has been incorporated into liposomes carrying vinorelbine, a potent anticancer drug, leading to enhanced systemic circulation, reduced hepatic clearance, and a significant increase in the drug’s half-life and area under the concentration-time curve (AUC). Similar improvements in pharmacokinetic profiles and enhanced anticancer potency have been observed with resveratrol-loaded TPGS-coated nanoparticles, suggesting a broad applicability across various therapeutic agents. These findings collectively underscore TPGS’s role as a multifunctional stabilizer, permeation enhancer, and performance booster in nanocarrier systems.
TPGS as a Functional Component of mRNA Delivery Systems
The advent of mRNA technology has revolutionized vaccine development and holds immense promise for treating a wide range of diseases. The ability of mRNA to encode virtually any antigen and its relatively rapid and scalable production make it an attractive therapeutic modality. However, the inherent instability of mRNA—susceptible to hydrolytic and oxidative degradation—necessitates sophisticated delivery systems to protect it from enzymatic breakdown and ensure efficient delivery into the cell cytoplasm for translation.
Nanoparticle-based delivery systems, particularly lipid nanoparticles (LNPs), have proven crucial for the successful clinical translation of mRNA vaccines, including those developed for COVID-19. LNPs shield mRNA from degradation and facilitate its entry into the cell. TPGS is emerging as a valuable component in these advanced delivery platforms. Its incorporation into nanoparticle and LNP formulations is expected to confer PEG-mediated "stealth" properties. This can reduce opsonization (the coating of particles by proteins that marks them for clearance by the immune system) and subsequent clearance by the reticuloendothelial system (RES), analogous to the function of PEG-lipids commonly used in LNPs.
The expected benefits include prolonged plasma half-life, increased AUC, reduced systemic clearance, and improved biodistribution. Furthermore, while TPGS itself is not an antioxidant, residual tocopherol impurities present in the mixture can scavenge reactive oxygen species (ROS). This antioxidant activity may further contribute to mRNA stability by mitigating oxidative damage, thereby reducing the formation of harmful byproducts and potentially lowering associated inflammatory responses and toxicity.
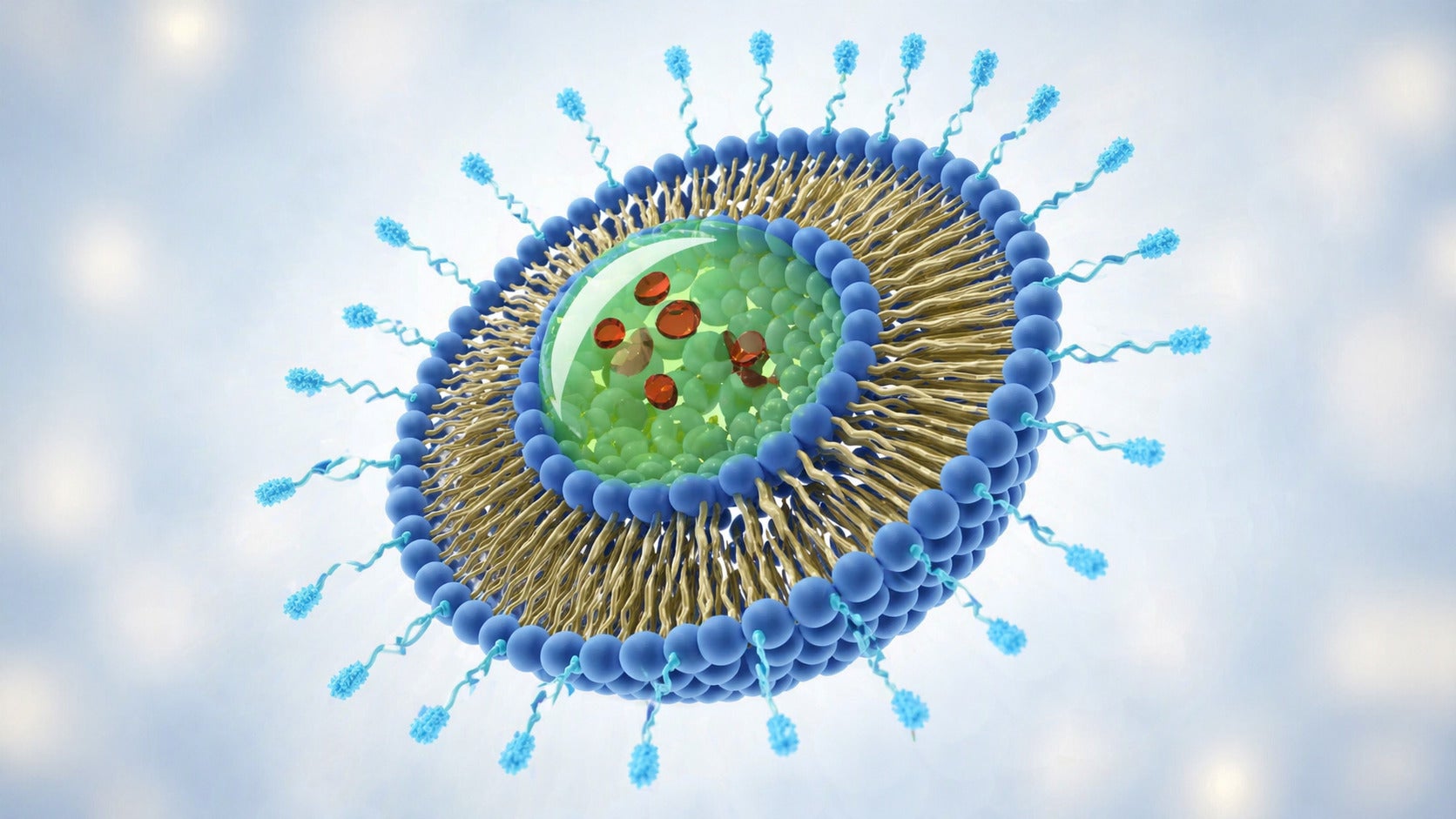
Beyond mRNA, TPGS has also shown promise in siRNA delivery. A novel biodegradable nanoparticle system composed of TPGS-b-poly(ε-caprolactone-ran-glycolide) was developed for siRNA targeting HIF-1α in nasopharyngeal carcinoma. These nanoparticles effectively delivered siRNA into cancer cells, demonstrating TPGS’s utility in gene therapy delivery systems.
Overcoming Major Challenges in Parenteral Development
The development of parenteral therapeutics is fraught with stringent requirements, including sterility, solubility, physical and chemical stability, biocompatibility, and patient tolerability. APIs with low aqueous solubility, susceptibility to degradation (hydrolysis, oxidation), and poor physical stability present significant formulation challenges. Issues such as precipitation, aggregation, and degradation during storage can compromise efficacy and safety. Moreover, ensuring excipient-API compatibility, container-closure integrity, endotoxin control, and minimizing particulate matter are paramount for patient safety.
Stability of Formulation
Maintaining the long-term stability of parenteral formulations, especially in aqueous solutions, is a critical hurdle. Physical instability can manifest as emulsion breakdown, particle growth, or drug precipitation, while chemical instability arises from degradation pathways that alter the drug or excipient structure. The formulation’s composition plays a pivotal role in determining overall stability. TPGS offers a robust solution. The ester linkage connecting d-α-tocopherol to the PEG-succinate moiety in TPGS exhibits remarkable stability under physiological conditions, with hydrolysis only occurring under extreme pH values. This inherent stability makes TPGS suitable for a wide range of parenteral formulations.
Delivery, Uptake, Formulation Functionality, and Sterility Considerations
Formulating nano-emulsions, lipid carriers, and nanoparticle systems requires a delicate balance between drug loading, particle size control, desired release kinetics, and the ability to maintain sterility. Terminal sterilization, a common method for many parenteral products, is not always feasible for heat-sensitive formulations. TPGS addresses these challenges by offering multiple functionalities. Its ability to stabilize emulsions, enhance drug solubility and loading, and form stable formulations that can withstand sterile filtration or terminal sterilization processes is invaluable. As illustrated in Table 3, TPGS presents significant advantages over common parenteral excipients like polysorbate 80 and Cremophor EL, which are associated with hypersensitivity reactions and infusion-related toxicities. Compared to Poloxamer 188, TPGS offers higher solubilizing efficiency and efflux inhibition. Its amphiphilic structure, which lacks in PEG alone, significantly improves drug encapsulation.
Safety Profile and Regulatory Acceptance
A crucial factor in the widespread adoption of any excipient, particularly in parenteral applications, is its safety profile and regulatory acceptance. TPGS boasts a well-established safety record. A recent evaluation by the European Food Safety Authority (EFSA) confirmed its favorable toxicological profile. Following intravenous administration, TPGS is rapidly distributed to highly perfused organs and undergoes ester hydrolysis to polyethylene glycol 1000 (PEG 1000), the primary metabolite. PEG 1000 is efficiently eliminated via urine and feces, minimizing the potential for accumulation. Toxicological studies in various animal species have shown no evidence of genotoxicity or treatment-related adverse effects, even at high doses. EFSA’s conclusion that TPGS poses no safety concern at proposed exposure levels further solidifies its suitability for pharmaceutical applications.
Conclusion: A Transformative Excipient for Modern Therapeutics
d-α-tocopheryl polyethylene glycol 1000 succinate (TPGS) is undergoing a significant transformation, evolving from a traditional oral excipient into a sophisticated, multifunctional component for advanced parenteral drug delivery. Its unique combination of properties—solubilization, steric stabilization, efflux pump inhibition, membrane interaction, and nanoparticle modulation—equips formulators with powerful tools to address long-standing challenges in delivering hydrophobic drugs, cytotoxic compounds, and increasingly complex nucleic acid therapeutics.
As the pharmaceutical industry continues to push the boundaries of innovation, with a growing emphasis on sophisticated injectable systems like mRNA-based therapies, lipid nanoparticles, nano-emulsions, and long-acting depots, TPGS is exceptionally positioned to play a transformative role. Its ability to enhance the safety, stability, and efficacy of these next-generation parenteral therapeutics marks it as an indispensable excipient for the future of medicine.
Authors’ Biographies:
MICHALIS NICOLAOU, PhD
Dr. Michalis Nicolaou is a distinguished scientific consultant and the President and CEO of Nicopharm Pharmaceutical Solutions. He also holds an adjunct professorship in Pharmaceutics at Western University College of Pharmacy. With a Ph.D. in Pharmaceutical Chemistry from the University of Kansas and over 30 years of experience in the pharmaceutical industry, his expertise spans product development, quality assurance, manufacturing, and regulatory affairs. Dr. Nicolaou has been instrumental in numerous projects focused on the design and optimization of vitamin E TPGS-based formulations.
ANDREAS M PAPAS, PhD
Dr. Andreas M. Papas is the CEO and a Board Member of Antares Health Products, Inc., and an adjunct professor in the College of Medicine at East Tennessee State University. A Fulbright Scholar, Dr. Papas is a recognized authority in the field of vitamin E and its applications. He is the author of the popular book "The Vitamin E Factor" and has authored numerous scientific papers and book chapters. Dr. Papas was a co-founder of YASOO Health, where he led the company in developing and commercializing innovative product concepts, supported by significant funding from the National Institutes of Health and the Cystic Fibrosis Foundation.
Antares Health Products Inc. is a premier supplier of vitamin E TPGS, a vital ingredient in the formulation of pharmaceuticals, dietary supplements, personal care products, food and beverages, and animal nutrition.
For additional information, please visit the Antares Health Products website at www.TPGS.com.
References:
- Paddon-Jones G. Emerging Applications of Vitamin E TPGS in Drug Delivery. Pharmaceutical Technology 46 (10) (2022).
- Robin Y. Using Tocophersolan for Drug Delivery. Pharm Technol. 2015 Jan 2;39(1):48–52.
- Papas AM. Vitamin E TPGS and its applications in nutraceuticals. In: Nutraceuticals. Elsevier; 2021. p. 991–1010.
- Nicolaou M, Surakitbanharn Y, OConnel S. MRTX1133 Pharmaceutical Compositions, US patent Application, US2025/0120981A1, 2025.
- Ahire E, Thakkar S, Darshanwad M, Misra M. Parenteral nanosuspensions: a brief review from solubility enhancement to more novel and specific applications. Acta Pharm Sin B. 2018 Sept;8(5):733–55.
- Li N, Mai Y, Liu Q, Gou G, Yang J. Docetaxel-loaded D-α-tocopheryl polyethylene glycol-1000 succinate liposomes improve lung cancer chemotherapy and reverse multidrug resistance. Drug Deliv Transl Res. 2021 Feb;11(1):131–41.
- Yang C, Wu T, Qi Y, Zhang Z. Recent Advances in the Application of Vitamin E TPGS for Drug Delivery. Theranostics. 2018;8(2):464–85.
- Guo Y, Luo J, Tan S, Otieno BO, Zhang Z. The applications of Vitamin E TPGS in drug delivery. Eur J Pharm Sci. 2013 May;49(2):175–86.
- Mu L, Feng SS. PLGA/TPGS Nanoparticles for Controlled Release of Paclitaxel: Effects of the Emulsifier and Drug Loading Ratio. Pharm Res. 2003 Nov;20(11):1864–72.
- Lambert KJ, Constantinides PP, Quay SC. Emulsion vehicle for poorly soluble drugs; United States patent US US6458373B1, 2002.
- Lakshmi, Singh S, Vijayakumar MR, Dewangan HK. Lipid Based Aqueous Core Nanocapsules (ACNs) for Encapsulating Hydrophillic Vinorelbine Bitartrate: Preparation, Optimization, Characterization and In vitro Safety Assessment for Intravenous Administration. Curr Drug Deliv. 2018 Sept 25;15(9):1284–93.
- Vijayakumar MR, Kosuru R, Singh SK, Prasad CB, Narayan G, Muthu MS, et al. Resveratrol loaded PLGA: d-α-tocopheryl polyethylene glycol 1000 succinate blend nanoparticles for brain cancer therapy. RSC Adv. 2016;6(78):74254–68.
- Vijayakumar MR, Kumari L, Patel KK, Vuddanda PR, Vajanthri KY, Mahto SK, et al. Intravenous administration of trans-resveratrol-loaded TPGS-coated solid lipid nanoparticles for prolonged systemic circulation, passive brain targeting and improved in vitro cytotoxicity against C6 glioma cell lines. RSC Adv. 2016;6(55):50336–48.
- Maurya L, Singh S, Rajamanickam VM, Narayan G. Vitamin E TPGS Emulsified Vinorelbine Bitartrate Loaded Solid Lipid Nanoparticles (SLN): Formulation Development, Optimization and In vitro Characterization. Curr Drug Deliv. 2018 Aug 16;15(8):1135–45.
- Vijayakumar MR, Vajanthri KY, Balavigneswaran CK, Mahto SK, Mishra N, Muthu MS, et al. Pharmacokinetics, biodistribution, in vitro cytotoxicity and biocompatibility of Vitamin E TPGS coated trans resveratrol liposomes. Colloids Surf B Biointerfaces. 2016 Sept;145:479–91.
- Cheng F, Wang Y, Bai Y, Liang Z, Mao Q, Liu D, et al. Research Advances on the Stability of mRNA Vaccines. Viruses. 2023 Mar 2;15(3):668.
- Schoenmaker L, Witzigmann D, Kulkarni JA, Verbeke R, Kersten G, Jiskoot W, et al. mRNA-lipid nanoparticle COVID-19 vaccines: Structure and stability. Int J Pharm. 2021 May;601:120586.
- Ingle RG, Fang WJ. An Overview of the Stability and Delivery Challenges of Commercial Nucleic Acid Therapeutics. Pharmaceutics. 2023 Apr 6;15(4):1158.
- Mehata AK, Setia A, Vikas V, Malik AK, Hassani R, Dailah HG, et al. Vitamin E TPGS-Based Nanomedicine, Nanotheranostics, and Targeted Drug Delivery: Past, Present, and Future. Pharmaceutics. 2023 Feb 21;15(3):722.
- Li X, Chen Y, Xu G, Zheng Y, Yan M, Li Z, et al. Nanoformulation of D-α-tocopheryl polyethylene glycol 1000 succinate-b-poly(ε-caprolactone-ran-glycolide) diblock copolymer for siRNA targeting HIF-1α for nasopharyngeal carcinoma therapy. Int J Nanomedicine. 2015 Feb;1375.
- Chaturvedi SH, Anand CM. A Detailed Concepts on Parenteral Preparation. Int J Pharm Res Appl. 2022;7(4):1883–95.
- ICH Q6A Specifications: Test Procedures and Acceptance Criteria for New Drug Substances and New Drug Products: Chemical Substances. [Internet]. International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH); 1999.
- ICH Q8(R2) Pharmaceutical Development [Internet]. International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH); 2009.
- Ren T, Li R, Zhao L, Fawcett JP, Sun D, Gu J. Biological fate and interaction with cytochromes P450 of the nanocarrier material, d-α-tocopheryl polyethylene glycol 1000 succinate. Acta Pharm Sin B. 2022 July;12(7):3156–66.















Leave a Reply