In an era defined by the dual crises of mounting environmental degradation and an urgent need for sustainable energy, a team of researchers at the University of Adelaide has unveiled a transformative approach to waste management. By harnessing the power of sunlight, scientists are developing sophisticated methods to convert discarded plastic into high-value fuels and industrial chemicals. This breakthrough, detailed in a comprehensive study led by PhD candidate Xiao Lu and Professor Xiaoguang Duan, represents a significant leap toward a circular economy, potentially turning one of the world’s most persistent pollutants into a vital energy resource.
The Global Plastic Crisis and the Energy Transition
The scale of the plastic problem is staggering. According to the Organization for Economic Co-operation and Development (OECD), global plastic production has more than doubled since the turn of the century, reaching approximately 460 million tonnes annually. Of this massive volume, less than 10% is successfully recycled. The remainder finds its way into landfills, incineration plants, or, most destructively, the natural environment. Estimates suggest that between 19 and 23 million tonnes of plastic waste leak into aquatic ecosystems every year, threatening biodiversity and entering the human food chain through microplastics.
Simultaneously, the global community is under immense pressure to decarbonize. The transition to a net-zero economy requires a fundamental shift away from fossil fuels toward "green" hydrogen and other carbon-neutral energy carriers. However, current methods of producing green hydrogen, such as water electrolysis, are energy-intensive and often expensive. The research emerging from the University of Adelaide suggests that plastic waste—rich in carbon and hydrogen—could provide a more efficient feedstock for fuel production than water alone, effectively killing two birds with one stone.
The Science of Solar-Driven Photoreforming
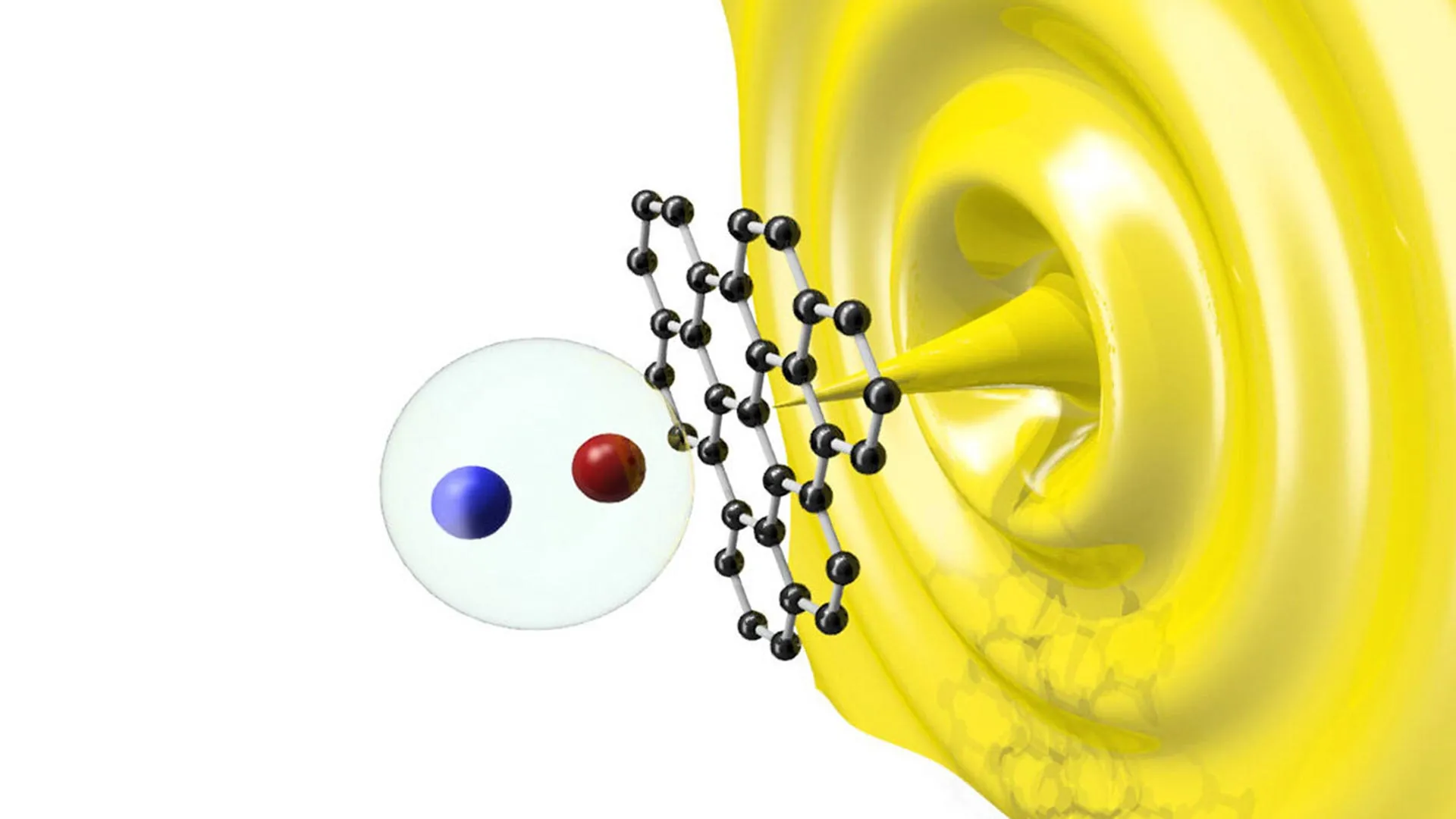
The core of this innovation lies in a process known as solar-driven photoreforming. Unlike traditional recycling, which involves melting and reforming plastic into lower-quality products, photoreforming uses light-sensitive materials, or photocatalysts, to drive chemical reactions at the molecular level.
When sunlight hits these photocatalysts—typically semiconductor materials—it creates electron-hole pairs. These highly reactive species interact with the polymer chains of the plastic and the surrounding medium (often water or an alkaline solution), breaking the carbon-carbon bonds. This process occurs at relatively low temperatures and atmospheric pressure, distinguishing it from gasification or pyrolysis, which require intense heat and significant energy input.
The primary outputs of this reaction are hydrogen gas (H2) and syngas (a mixture of hydrogen and carbon monoxide). Hydrogen is widely regarded as the fuel of the future, particularly for heavy industry and long-haul transport, because its only byproduct at the point of use is water vapor. Furthermore, the photoreforming process can be "tuned" to produce specific organic chemicals, such as acetic acid—a key ingredient in the production of textiles, plastics, and food preservatives—and even diesel-range hydrocarbons.
Comparative Efficiency and Thermodynamic Advantages
One of the most compelling arguments for plastic-to-fuel conversion is its thermodynamic efficiency compared to traditional green hydrogen production. In standard water splitting, the oxygen evolution reaction (OER) is the "bottleneck"—it is a slow, energy-demanding process.
"Plastics are essentially solid hydrocarbons," explains the research team. Because the chemical bonds in polymers like polyethylene (PE) or polypropylene (PP) are easier to oxidize than water molecules, the energy barrier for the reaction is significantly lower. By replacing the water oxidation step with plastic oxidation, the system can produce hydrogen at a much higher rate using the same amount of solar energy. This "sacrificial" use of plastic not only cleans up the environment but also reduces the cost per kilogram of hydrogen produced, making solar-to-fuel technology more economically competitive with fossil-fuel-derived hydrogen.
Chronology of Development and Experimental Milestones
The journey toward solar-powered plastic conversion has evolved over several decades, moving from theoretical chemistry to practical laboratory application.
- 2010s: Early experiments focused on the degradation of organic pollutants in water. Researchers began to realize that the same principles could be applied to larger polymer chains.
- 2017–2019: Proof-of-concept studies demonstrated that pure, laboratory-grade plastics could be converted into hydrogen under UV light. However, the efficiency remained low, and the catalysts were expensive.
- 2020–2022: Research shifted toward "visible light" photocatalysis, allowing the technology to use the full spectrum of sunlight rather than just UV rays. This period saw the introduction of more robust catalysts, such as carbon nitrides and metal-organic frameworks (MOFs).
- 2023–2024: The Adelaide University study, published in Chem Catalysis, represents the current state-of-the-art. The team moved beyond pure plastics to test real-world waste, achieving continuous operation for over 100 hours. This milestone is crucial for demonstrating the stability required for industrial scaling.
In recent trials, the team reported not only high yields of hydrogen but also the successful synthesis of value-added liquid chemicals. The ability to produce acetic acid and diesel-range molecules simultaneously with hydrogen increases the "atom economy" of the process, ensuring that almost every part of the plastic waste is converted into something useful.
Addressing the Complexity of Real-World Waste
While laboratory results are promising, the transition to industrial application faces significant hurdles. The primary challenge is the heterogeneity of plastic waste. Unlike the pure pellets used in early research, consumer waste is a "cocktail" of different polymers (PE, PET, PVC, PS), each with different chemical properties.
Furthermore, commercial plastics are rarely pure. They contain a variety of additives, including:
- Colorants and Dyes: These can block sunlight from reaching the photocatalyst.
- Stabilizers: Designed to prevent the plastic from breaking down, these chemicals can inhibit the photoreforming reaction.
- Flame Retardants and Plasticizers: These may introduce impurities into the final fuel or poison the catalyst.
Professor Xiaoguang Duan emphasizes that efficient pre-treatment and sorting are essential. "We cannot simply throw a mixed bag of trash into a reactor and expect high-quality fuel," he noted. "The future of this technology depends on integrated waste management systems where sorting and chemical processing work hand-in-hand."
Technical Hurdles and Catalyst Durability
Another critical area of focus is the longevity of the photocatalysts. In a laboratory setting, catalysts are often tested for short durations. However, for a plant to be commercially viable, these materials must remain active for thousands of hours under harsh chemical conditions.
Current catalysts can suffer from "photocorrosion" or become "poisoned" by the byproducts of the plastic breakdown. Researchers are currently exploring "heterojunction" catalysts—composites of two or more materials—that are more durable and efficient at capturing solar energy. There is also a push to use earth-abundant materials like iron, copper, and carbon, rather than precious metals like platinum or ruthenium, to keep costs low.
Engineering the Reactors of Tomorrow
Beyond the chemistry, the engineering of the reactors presents its own set of challenges. Traditional batch reactors, where waste is processed in "lots," are often inefficient for large-scale production. The University of Adelaide team is looking toward continuous-flow reactors. In these systems, a slurry of plastic waste and catalysts flows through transparent tubes exposed to sunlight, allowing for a steady output of fuel.
Additionally, the separation of the final products remains energy-intensive. When the reactor produces a mixture of hydrogen, methane, and CO2, as well as liquid acetic acid, these must be purified. If the energy required to separate these chemicals exceeds the energy stored in the produced fuel, the environmental benefit is lost. To mitigate this, researchers are designing "membrane reactors" that can separate hydrogen in real-time as it is produced.
Stakeholder Perspectives and Economic Implications
The potential for solar-powered plastic-to-fuel technology has caught the attention of various stakeholders, from environmental NGOs to the petrochemical industry.
Environmental advocates, while generally supportive, caution against "greenwashing." Organizations like Greenpeace have historically argued that the focus should remain on reducing plastic production rather than finding ways to "burn" or convert it. However, many circular economy experts argue that since 6.3 billion tonnes of plastic waste already exist on the planet, chemical upcycling is a necessary pragmatic solution.
From an industrial perspective, major energy companies are increasingly interested in "waste-to-X" technologies. As carbon taxes become more prevalent globally, the ability to turn a waste liability into a fuel asset offers a significant financial incentive. Analysts suggest that if the technology can reach a "Levelized Cost of Hydrogen" (LCOH) comparable to fossil-fuel-based methods, it could disrupt the multi-billion dollar waste management and energy sectors.
Policy and the Path to 2050
The success of these technologies is inextricably linked to global policy frameworks. The United Nations is currently negotiating a legally binding global treaty to end plastic pollution. Such a treaty could mandate the adoption of advanced recycling technologies and provide the subsidies necessary to move solar photoreforming from the lab to the pilot plant.
In Australia, the government’s "National Hydrogen Strategy" and "National Plastics Plan" provide a supportive policy environment for such innovations. By positioning itself as a leader in green hydrogen, Australia could export not just the fuel, but the proprietary technology required to produce it from waste.
Conclusion: A Vision for a Sustainable Future
The research led by Xiao Lu and Professor Xiaoguang Duan at the University of Adelaide provides a roadmap for a future where plastic is no longer a symbol of environmental failure, but a cornerstone of the clean energy transition. By utilizing the most abundant energy source on Earth—the sun—to dismantle the most stubborn waste product of the 20th century, scientists are redefining the limits of sustainability.
While significant engineering and chemical challenges remain, the progress made in hydrogen yield and catalyst stability is undeniable. Over the next decade, the focus will shift to pilot-scale demonstrations and the integration of these reactors into existing waste infrastructure. If successful, solar-powered plastic-to-fuel conversion will play a pivotal role in achieving a low-carbon, circular economy, proving that with enough innovation, today’s pollution can indeed become tomorrow’s power.













Leave a Reply