In a landmark achievement for regenerative medicine and oncology, a multidisciplinary team of scientists at the University of Basel and University Hospital Basel has successfully engineered a complex, functional model of human bone marrow using exclusively human cells. This breakthrough, published in the prestigious journal Cell Stem Cell, represents the first time that the intricate "blood factory" of the human body has been replicated in a laboratory setting without the use of animal-derived components or inter-species scaffolding. Led by Professor Ivan Martin and Dr. Andrés García-García from the Department of Biomedicine, the research offers a transformative tool for studying hematological diseases, testing new pharmaceutical compounds, and potentially pioneering personalized treatment protocols for patients suffering from blood-related malignancies.
The Architectural Complexity of the Human Blood Factory
The human bone marrow is not merely a passive reservoir for blood cells; it is a highly specialized and dynamic tissue network. It functions as a sophisticated ecosystem where bone cells, nerves, blood vessels, and various immune cells interact to regulate the production of billions of new blood cells every day. This process, known as hematopoiesis, occurs within specialized microenvironments called "niches."
The Basel researchers focused their efforts on replicating the "endosteal niche," a specific region located near the inner surface of the bone. This niche is of paramount importance to medical science because it is believed to be the site where hematopoietic stem cells (HSCs) reside and where certain types of cancer, such as leukemia, find refuge. In many cases of blood cancer, malignant cells utilize the protective environment of the endosteal niche to resist chemotherapy, leading to disease relapse. By successfully recreating this niche, the researchers have provided a window into a previously inaccessible theater of human biology.
Methodology: From Mineral Scaffolds to Pluripotent Stem Cells
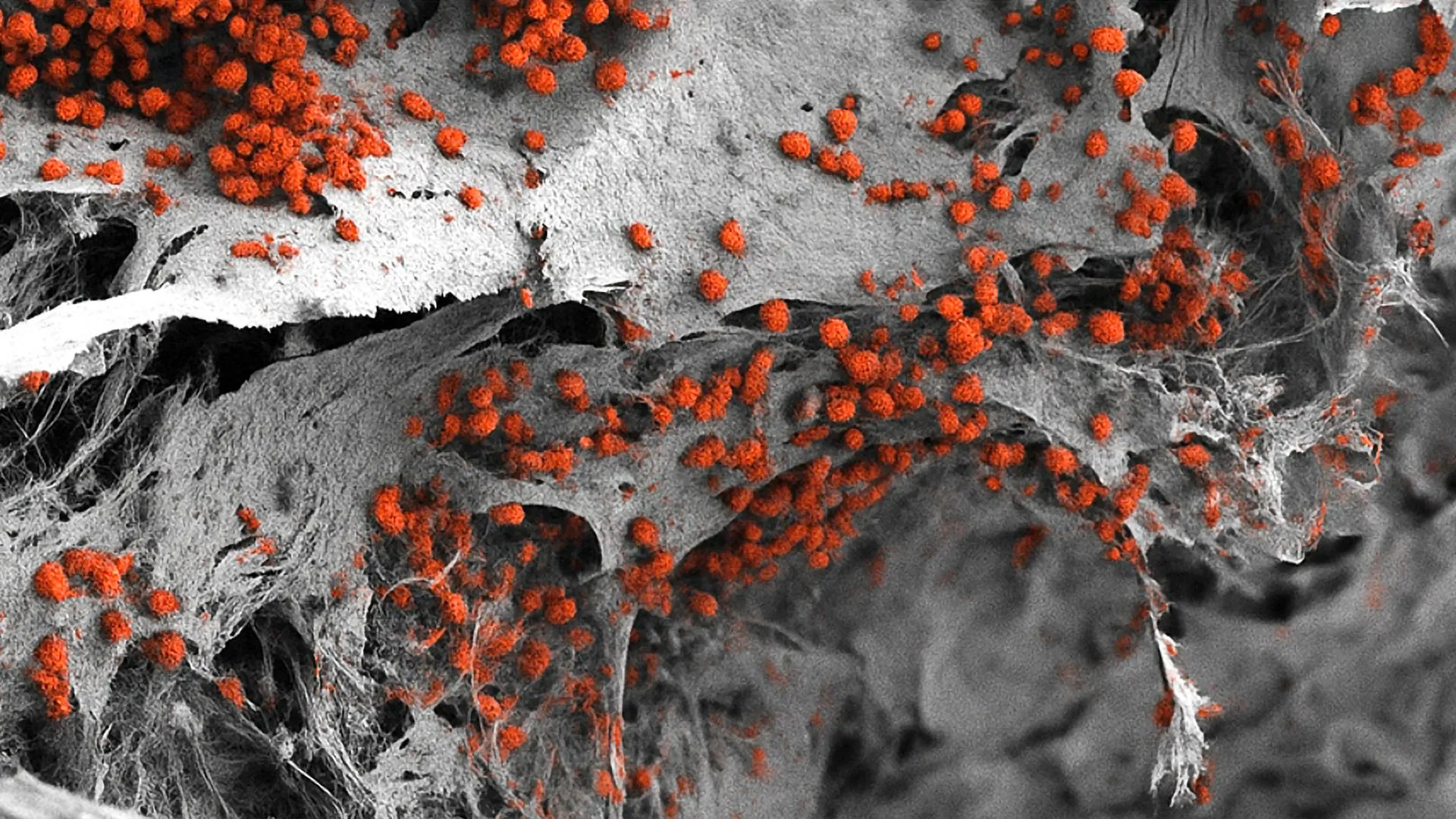
The creation of this three-dimensional model required a sophisticated blend of materials science and molecular biology. The foundation of the model is a synthetic framework made of hydroxyapatite. Hydroxyapatite is a naturally occurring mineral form of calcium apatite that constitutes the primary inorganic component of human bone and tooth enamel. By using this material, the team provided a bio-mimetic physical structure that mirrors the stiffness and chemical composition of actual human bone.
To populate this scaffold, the team utilized human induced pluripotent stem cells (iPSCs). These are adult cells—often skin or blood cells—that have been genetically "reprogrammed" back into an embryonic-like state using specific molecular signals, a technique famously pioneered by Shinya Yamanaka. Because iPSCs have the potential to differentiate into any cell type in the body, the Basel researchers were able to guide them through a series of controlled developmental stages.
Over several weeks, these stem cells transformed into a diverse array of bone marrow constituents, including osteoblasts (bone-forming cells), endothelial cells (which form blood vessels), and stromal cells. The result was a living, breathing 3D structure that reached dimensions of eight millimeters in diameter and four millimeters in thickness—a significant scale-up from previous micro-scale laboratory models.
A Chronology of Bone Marrow Research
The journey to this discovery is the culmination of decades of incremental progress in hematology and bioengineering.
- The 1960s: Ernest McCulloch and James Till first identified the existence of hematopoietic stem cells, proving that the bone marrow contained "mother cells" capable of regenerating the entire blood system.
- The 1990s: Researchers began developing two-dimensional (2D) cell culture systems. While useful, these flat environments failed to capture the three-dimensional interactions and physical pressures found in living tissue.
- The 2000s: The advent of "Organ-on-a-Chip" technology allowed for better nutrient flow and mechanical stimulation, but these systems often relied on mouse cells or simplified human cell lines that did not represent the full complexity of the marrow.
- 2010-2020: Progress in 3D printing and synthetic biology allowed for more complex scaffolds, yet the integration of multiple cell types (nerves, vessels, and bone) remained an elusive goal.
- 2024: The Basel team’s publication in Cell Stem Cell marks the shift to fully humanized, multi-component 3D systems that can sustain blood cell formation for extended periods in a laboratory environment.
Data and Performance: Maintaining Life in Vitro
One of the most significant findings of the study was the model’s ability to maintain human blood cell formation for several weeks. In previous attempts to culture human bone marrow, the hematopoietic stem cells often lost their "stemness"—their ability to self-renew—very quickly, or they differentiated into mature blood cells too rapidly, exhausting the supply.
In the Basel model, the researchers observed that the synthetic endosteal niche provided the necessary biochemical and physical cues to keep the stem cells in a healthy, functional state. Analysis of the 3D structure showed that the spatial organization of the cells closely matched that of native human tissue. This longevity is crucial for long-term drug testing and for observing the slow progression of chronic blood diseases.
Implications for the "3Rs" and Animal Welfare
A primary driver behind this research is the global scientific push toward the "3Rs": the Replacement, Reduction, and Refinement of animal experiments. For decades, the gold standard for bone marrow research has been the mouse model. While mice share many genetic similarities with humans, their hematological systems differ in fundamental ways, particularly in how they respond to certain inflammatory signals and how their immune cells interact with cancer.
Professor Ivan Martin emphasized that while mouse studies have provided the foundation of our current knowledge, they are not perfect proxies for human biology. "Our model brings us closer to the biology of the human organism," Martin stated. By using a system derived entirely from human cells, researchers can bypass the physiological discrepancies between species. This not only improves the accuracy of the data but also aligns with the University of Basel’s ethical commitment to reducing the number of animals used in laboratory research.
Toward Personalized Medicine in Oncology
Perhaps the most exciting application of this technology lies in the realm of personalized medicine. Currently, patients with blood cancers like leukemia or multiple myeloma often undergo a "trial and error" approach to chemotherapy, where doctors prescribe standard treatments and wait to see if the patient responds.
The Basel team envisions a future where a patient’s own cells are used to create a "personalized bone marrow model" in the lab. Doctors could then introduce various combinations of drugs to this specific model to see which therapy most effectively kills the cancer cells while sparing the healthy ones. This would allow for the tailoring of treatment to the unique genetic profile of the individual, potentially increasing survival rates and reducing the side effects of ineffective treatments.
Challenges and Future Directions
Despite the breakthrough, there are hurdles to overcome before this model becomes a standard tool in every hospital. Dr. Andrés García-García noted that the current size of the model—while impressive from a biological standpoint—is actually a drawback for high-throughput drug testing. Pharmaceutical companies often need to test thousands of different chemical compounds simultaneously.
"For this specific purpose, the size of our bone marrow model might be too large," García-García explained. The next phase of research will likely involve miniaturizing the system into "micro-niches" that can be arrayed on plates for automated testing. Additionally, while the model includes bone cells and blood vessels, the integration of a fully functional nervous system within the marrow remains a complex challenge for future iterations.
Broader Impact on Biotechnology and Public Health
The success of the Basel study is expected to resonate across the biotechnology sector. As the cost of drug development continues to rise—often exceeding $2 billion to bring a single drug to market—the industry is desperate for more predictive models. High failure rates in clinical trials are often attributed to the fact that drugs that work in mice fail to show efficacy or safety in humans. A fully human bone marrow model could filter out these failures much earlier in the development pipeline, saving billions of dollars and years of research time.
Furthermore, the study highlights the growing importance of "organoids" and synthetic tissues in the modern medical landscape. As researchers become more adept at growing human organs and tissues in vitro, the boundary between laboratory benchwork and clinical application continues to blur.
Conclusion
The creation of a fully human, 3D bone marrow model by the University of Basel represents a paradigm shift in how we study the core of our circulatory and immune systems. By combining hydroxyapatite scaffolds with the versatility of induced pluripotent stem cells, the research team has moved beyond the limitations of animal models and 2D cultures.
As this technology matures, it promises to provide deeper insights into the mechanisms of blood cancer, more efficient paths for drug discovery, and a new era of personalized healthcare. While the bone marrow may "operate quietly" within our bodies, the noise generated by this scientific advancement is likely to be felt across the global medical community for years to come. The study not only underscores the power of human-centric research but also reaffirms the potential of bioengineering to solve some of the most complex puzzles in human biology.















Leave a Reply