A multidisciplinary research team from the Department of Medicine at the School of Clinical Medicine, LKS Faculty of Medicine, the University of Hong Kong (HKUMed), has achieved a significant breakthrough in skeletal biology by identifying the specific biological process that allows physical activity to maintain and enhance bone density. The study, published in the prestigious international journal Signal Transduction and Targeted Therapy, pinpointed a protein known as Piezo1 as the body’s internal "exercise sensor" within the bone marrow. This discovery provides a long-sought-after molecular explanation for why weight-bearing exercise is essential for bone health and, more importantly, provides a blueprint for pharmacological interventions that could replicate these benefits for patients who are physically unable to exercise.
The implications of this research are far-reaching, particularly for the treatment of osteoporosis and other age-related bone loss conditions. By decoding the signaling pathways that convert mechanical pressure into bone growth, the HKUMed team has opened the door to the development of "exercise mimetics." These are therapeutic compounds designed to "trick" the skeletal system into activating its growth and repair mechanisms, potentially offering a lifeline to the elderly, the bedridden, and those suffering from chronic illnesses that limit mobility.
The Global and Local Burden of Osteoporosis
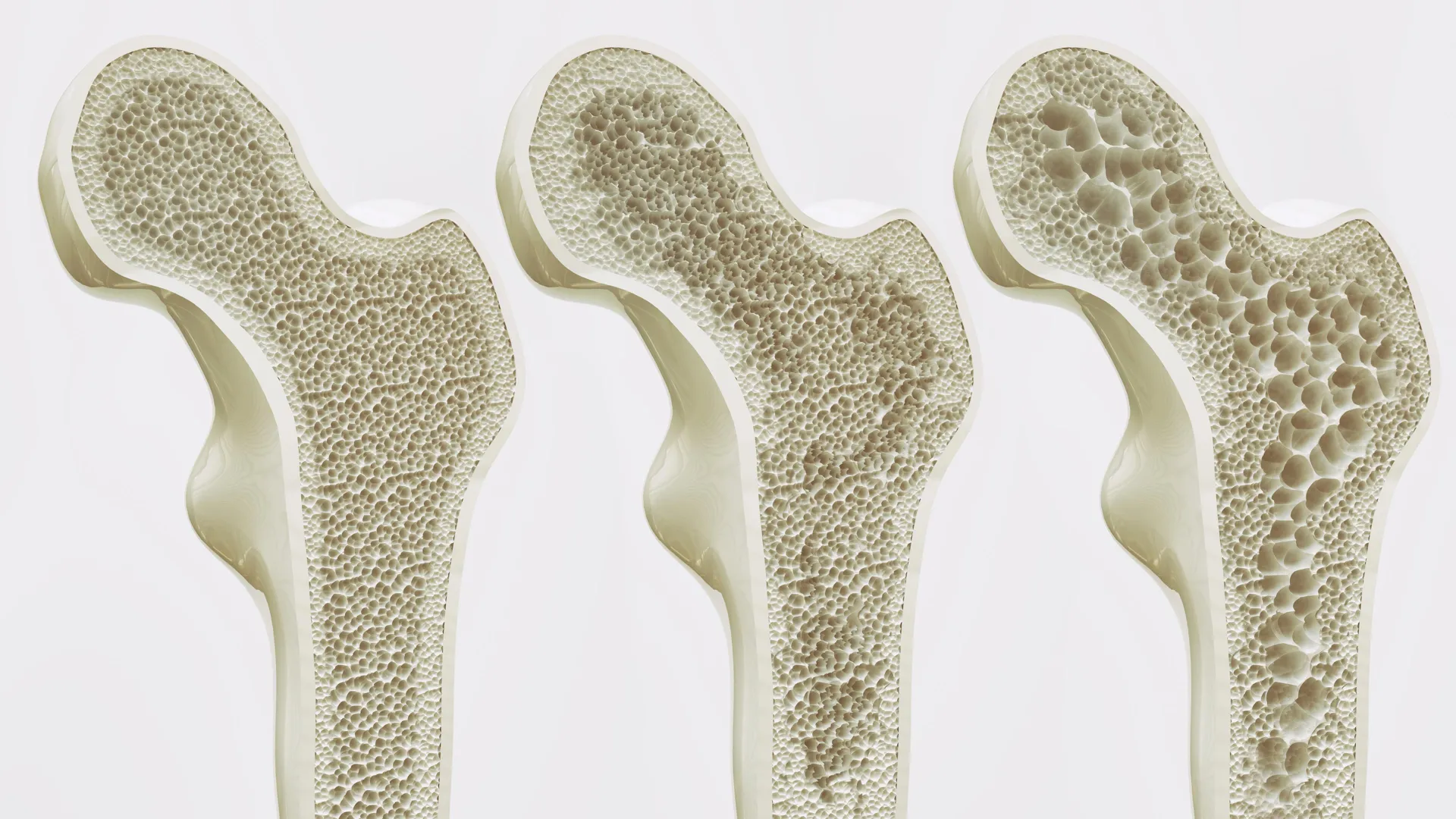
To understand the magnitude of this discovery, it is necessary to consider the current state of bone health globally. Osteoporosis is a systemic skeletal disorder characterized by low bone mass and micro-architectural deterioration of bone tissue, leading to increased bone fragility and a consequent increase in fracture risk. It is often referred to as a "silent epidemic" because bone loss occurs without symptoms until a fracture happens.
According to data from the World Health Organization (WHO), osteoporosis affects hundreds of millions of people worldwide. Statistics indicate that approximately one in three women and one in five men over the age of 50 will suffer an osteoporotic fracture in their remaining lifetime. These injuries, particularly hip and vertebral fractures, are associated with high rates of morbidity and mortality. In many cases, a hip fracture leads to a permanent loss of independence, with nearly 25% of patients requiring long-term nursing home care following the injury.
In Hong Kong, the challenge is exacerbated by a rapidly aging population. Estimates suggest that osteoporosis affects 45% of women and 13% of men aged 65 and older in the territory. As the demographic shift continues, the strain on the public healthcare system is expected to grow exponentially. Current therapeutic options, while effective for many, often come with side effects or require the patient to maintain an active lifestyle to maximize the drug’s efficacy. For those who are already frail or immobilized, the current medical toolkit is notably limited.
The Biological Mechanism: Bone versus Fat
The core of the HKUMed study focuses on the behavior of mesenchymal stem cells (MSCs) located within the bone marrow. These versatile cells serve as the progenitors for various tissues; they have the potential to differentiate into osteoblasts (bone-forming cells) or adipocytes (fat cells). In a healthy, active individual, there is a delicate balance in this differentiation process, heavily favored toward the production of bone tissue due to the mechanical stimuli provided by daily movement and exercise.
However, as the body ages, or when physical activity decreases significantly, this balance undergoes a detrimental shift. The MSCs increasingly differentiate into fat cells rather than bone cells. This "fatty infiltration" of the bone marrow is a hallmark of aging and osteoporosis. Not only does it result in less new bone being formed, but the accumulation of marrow adipose tissue (MAT) is also thought to create an environment that further inhibits bone regeneration and weakens the existing skeletal structure.
Until now, the exact molecular "switch" that tells these stem cells to become bone instead of fat in response to physical force remained elusive. The HKUMed team, led by Professor Xu Aimin, sought to identify the mechanical sensor responsible for this decision-making process.
Piezo1: The Molecular Exercise Sensor
Through a series of sophisticated experiments involving mouse models and human stem cell cultures, the researchers identified Piezo1 as the primary mechanosensor in bone marrow MSCs. Piezo proteins are a class of ion channels that respond to mechanical pressure by allowing the flow of ions into cells, thereby triggering various biological responses. The importance of the Piezo family was highlighted globally when the 2021 Nobel Prize in Physiology or Medicine was awarded for the discovery of these receptors.
The HKUMed study demonstrated that when Piezo1 is activated by the mechanical forces generated during exercise—such as tension, compression, and fluid shear stress—it sends signals that promote the expression of bone-forming genes while simultaneously suppressing the pathways that lead to fat formation.
In laboratory models where Piezo1 was genetically removed from MSCs, the researchers observed a dramatic acceleration of bone loss and a massive increase in bone marrow fat, even when the subjects were active. Conversely, when Piezo1 was chemically or mechanically stimulated, bone density was maintained or even increased. The study further revealed that a lack of Piezo1 activity triggers the release of specific inflammatory signals, namely Ccl2 and lipocalin-2. These "bad" signals create a pro-inflammatory environment that pushes stem cells toward fat production and actively interferes with bone growth. By blocking these specific signals, the researchers were able to partially restore healthy bone formation, suggesting multiple targets for future drug development.
A New Era of Exercise Mimetics
The concept of "exercise mimetics" is one of the most exciting frontiers in modern pharmacology. While the idea of a "pill that replaces the gym" is often used in a sensationalist context, for clinical populations, it represents a necessary medical intervention.
Professor Xu Aimin, Director of the State Key Laboratory of Pharmaceutical Biotechnology and Chair Professor in the Department of Medicine at HKUMed, emphasized that the goal is not to replace exercise for the healthy, but to provide the biological benefits of movement to those who are physically prevented from achieving them. "Current treatments rely heavily on physical activity, which many patients simply cannot perform," Professor Xu stated. "We need to understand how our bones get stronger when we move before we can find a way to replicate those benefits at the molecular level. This study is a critical step toward that goal."
Dr. Wang Baile, Research Assistant Professor at HKUMed and co-leader of the study, highlighted the potential for Piezo1-targeting drugs to transform geriatric care. By chemically activating the Piezo1 pathway, doctors could potentially maintain the bone mass of bedridden patients or those recovering from major surgery, preventing the rapid skeletal decay that usually accompanies prolonged immobilization.
Collaborative Research and Global Impact
The study was a highly collaborative effort, involving experts from across the globe. Professor Eric Honoré, a team leader at the Institute of Molecular and Cellular Pharmacology of the French National Centre for Scientific Research (CNRS), played a pivotal role as a co-leader. His expertise in mechanobiology helped bridge the gap between mechanical stimulus and molecular response.
"This offers a promising strategy beyond traditional physical therapy," noted Professor Honoré. "In the future, we could potentially provide the biological benefits of exercise through targeted treatments, thereby slowing bone loss in vulnerable groups and substantially reducing their risk of fractures."
The research was supported by a wide array of funding bodies, reflecting its international importance. These include the Research Grants Council of Hong Kong, the Health Bureau of the HKSAR Government, the National Key R&D Program of China, and various French national research agencies. This level of support underscores the global consensus that finding a solution to age-related bone loss is a public health priority.
Chronology and Future Directions
The journey to this discovery involved several years of incremental research:
- Initial Observation: Researchers noted the inverse relationship between bone marrow fat and bone density in aging populations.
- Hypothesis Formulation: The team hypothesized that a mechanosensor must exist on MSCs to dictate their differentiation path.
- Protein Screening: Extensive screening identified Piezo1 as a highly expressed mechanosensor in bone-forming regions.
- Validation: Using CRISPR technology and mouse models, the team validated that the absence of Piezo1 led to osteoporosis-like symptoms.
- Signal Mapping: The identification of Ccl2 and lipocalin-2 as the downstream effectors of Piezo1 deficiency.
- Publication: The final findings were peer-reviewed and published in 2024.
The research team is now moving toward the next phase of the project: clinical translation. This involves identifying small-molecule compounds that can safely and effectively activate Piezo1 in humans. While the transition from animal models to human clinical trials is complex and can take several years, the identification of the Piezo1-Ccl2-lipocalin-2 axis provides a clear map for drug developers.
Analysis of Implications for Healthcare Policy
If successful, the development of Piezo1-based therapies could revolutionize the economic landscape of elderly care. The direct costs of treating fractures—surgery, hospitalization, and rehabilitation—are staggering. Indirect costs, including lost productivity for caregivers and the long-term social costs of disability, are even higher.
By shifting the focus from treating fractures to preventing the underlying bone decay through molecular mimicry of exercise, healthcare systems could see a significant reduction in the incidence of "fragility fractures." This proactive approach aligns with the global shift toward "healthy aging" initiatives, where the goal is not just to extend life, but to extend the period of life spent in good health and independence.
The HKUMed study stands as a testament to the power of molecular biology in solving structural human problems. By decoding the language of movement used by our cells, researchers have moved one step closer to ensuring that the loss of mobility does not inevitably lead to the loss of skeletal integrity. For the millions of people currently facing the limitations of age or illness, the "exercise sensor" discovery offers a new horizon of hope for a future of stronger bones and greater independence.















Leave a Reply