The human body is a symphony of electrical activity, a constant flow of microscopic charges that orchestrate life’s fundamental processes. Every second, these invisible signals facilitate communication between cells, drive energy production, and underpin metabolism. This intricate dance of charge transport, particularly the controlled movement of protons – positively charged hydrogen ions – across and within cell membranes, acts as a sophisticated biological control system. Understanding these mechanisms is not only crucial for deciphering biological functions but also holds significant promise for advancing technologies like batteries and fuel cells, where efficient proton conduction is paramount.
At the heart of this vital charge transport are phosphoric acid (H₃PO₄) and its related compounds. These ubiquitous molecules are fundamental building blocks of life, forming the backbone of DNA and RNA, integral components of cell membranes, and central to adenosine triphosphate (ATP), the universal energy currency of cells. Their remarkable ability to facilitate the movement of protons has long been recognized, but the precise molecular choreography of this process has remained an elusive puzzle.
A Decade-Long Quest to Understand Proton Shuttling
For years, scientists have acknowledged that protons don’t simply wander freely through phosphate-rich environments. Instead, they engage in a process known as "proton-shuttling," a rapid relay race where protons hop from one molecule to another, guided by a network of hydrogen bonds. This efficient mechanism allows for swift charge transfer, a critical factor in biological and technological applications.
However, the exact molecular configurations and the initial steps of this proton transfer have been a subject of intense scientific scrutiny. This quest for clarity has involved a multi-national collaboration, spearheaded by researchers from the Department of Molecular Physics at the Fritz Haber Institute in Germany, in conjunction with esteemed colleagues from Leipzig University and institutions across the United States. Their collective efforts have been dedicated to dissecting the earliest stages of proton transfer within a key molecular structure.
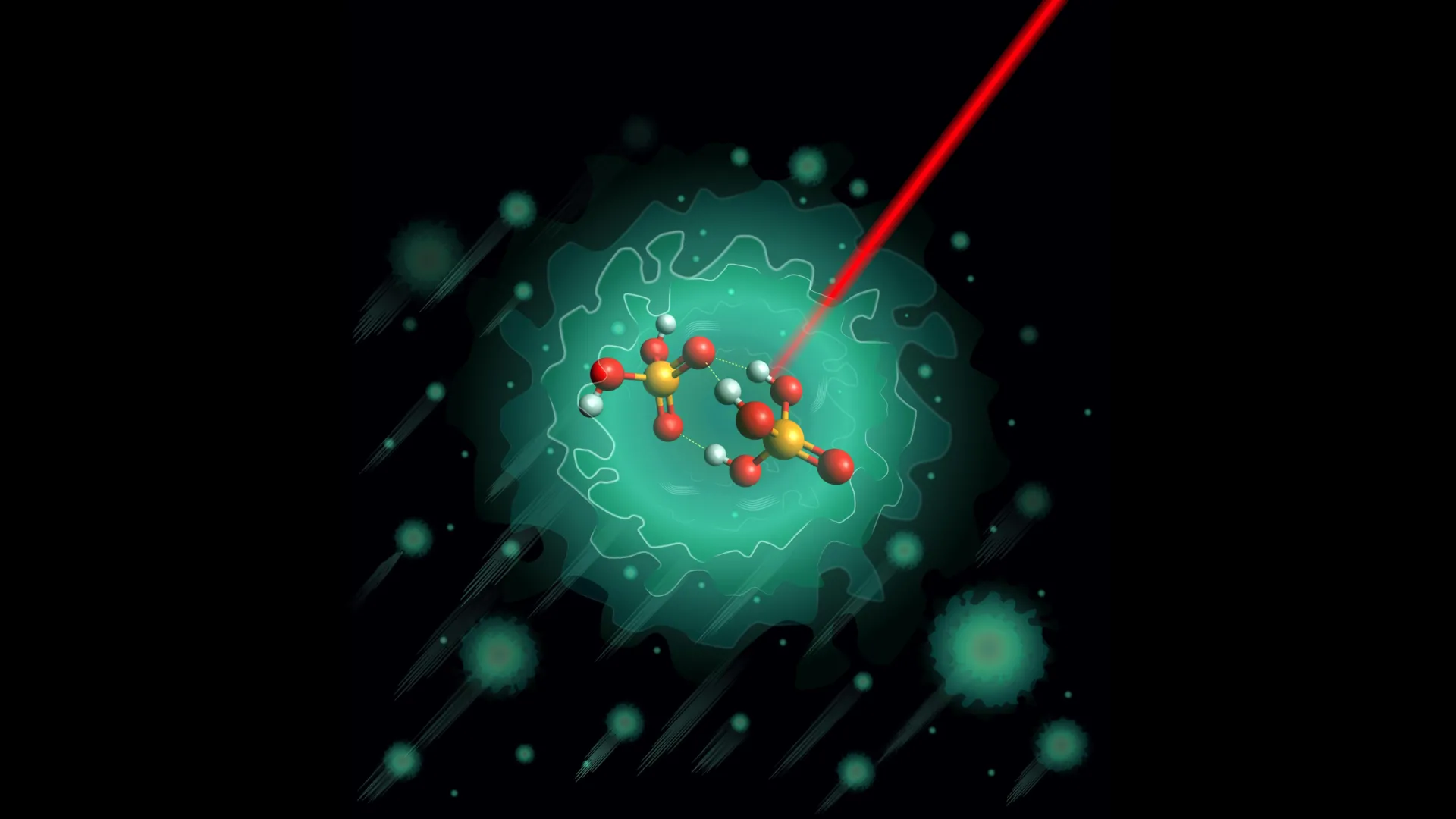
The Unveiling of a Fundamental Anion: H₃PO₄·H₂PO₄⁻
Previous theoretical models had posited that a specific negatively charged molecule, the deprotonated dimer H₃PO₄·H₂PO₄⁻, plays a pivotal role in initiating the proton-shuttling cascade. This anionic dimer, essentially a molecule of phosphoric acid that has lost a proton and bonded with another phosphoric acid molecule that has gained one, was identified as a prime candidate for detailed investigation. Its structure, and the way it facilitates proton movement, was believed to hold the key to understanding the broader proton transport mechanisms.
Probing the Depths of Molecular Motion at Extreme Cold
To overcome the inherent complexities and transient nature of molecular interactions, the research team employed an innovative approach. They meticulously synthesized the H₃PO₄·H₂PO₄⁻ dimer in a controlled laboratory setting. The true breakthrough came with their decision to cool these fragile molecules to an astonishingly low temperature: just 0.37 degrees Celsius above absolute zero (-273.15 °C or -459.67 °F).
This extreme cooling was achieved by encasing the molecules within helium nanodroplets. These minuscule, superfluid droplets act as near-perfect cryogenic cradles, effectively freezing out all but the most fundamental molecular vibrations and interactions. At such frigid temperatures, the researchers could effectively isolate and stabilize the target dimer, minimizing unwanted disturbances that would typically obscure precise structural analysis.
The stabilized dimer was then subjected to sophisticated infrared spectroscopy. This technique, which probes the vibrational modes of molecules, provides detailed fingerprints of their structure and bonding. By analyzing the specific frequencies of infrared light absorbed by the H₃PO₄·H₂PO₄⁻ dimer, the scientists could infer its precise atomic arrangement.
Crucially, these experimental findings were not analyzed in isolation. They were integrated with advanced quantum chemical calculations. These computational methods, rooted in the principles of quantum mechanics, allow scientists to predict molecular geometries, energies, and reaction pathways. By juxtaposing the experimental data with theoretical predictions, the research team was able to achieve an unprecedented level of clarity regarding the dimer’s structure. This synergistic approach, combining empirical observation with theoretical insight, proved far more powerful than either method could offer alone.
A Singular Structure Emerges: Challenging Theoretical Preconceptions
The results of this rigorous investigation yielded a surprising and significant finding. Leading theoretical models had predicted that the H₃PO₄·H₂PO₄⁻ dimer could exist in two distinct, equally probable structural configurations. These models suggested a degree of flexibility or dynamic equilibrium between these forms.
However, the high-precision infrared spectroscopy data painted a different picture. The experimental evidence unequivocally pointed to the existence of only one stable structural configuration for the dimer under the studied conditions. This single, well-defined structure revealed a relatively rigid molecular arrangement.
Key Structural Features Revealed:
- Hydrogen Bond Network: The stable structure features a tightly woven network of three strong hydrogen bonds. These bonds are not arranged in a linear fashion but rather converge around a shared oxygen atom, creating a compact and stable core.
- Proton Mobility Barriers: This specific arrangement of hydrogen bonds and the overall molecular conformation create significant energy barriers. These barriers effectively restrict the free movement of protons within the dimer, suggesting a more controlled and perhaps localized proton transfer mechanism than initially anticipated by some models.
- Ubiquitous Bonding Pattern: Notably, similar hydrogen bonding patterns have been observed in other, larger clusters of phosphoric acid molecules. This observation strongly suggests that this specific structural motif, characterized by the three hydrogen bonds converging on a shared oxygen atom, may be a recurrent and fundamental feature in how phosphoric acid and its derivatives organize themselves and interact.
This discrepancy between theoretical predictions and experimental observations underscores a critical point in scientific inquiry: even the most sophisticated theoretical models are not infallible. They are approximations of reality, and experimental verification remains an indispensable cornerstone for confirming, refining, or even overturning established theoretical frameworks. The precision achieved in this study highlights the power of advanced experimental techniques in revealing subtle molecular details that can elude purely theoretical approaches.
Implications: Illuminating Nature’s Proton Highway and Beyond
The discovery of a single, well-defined structure for the H₃PO₄·H₂PO₄⁻ dimer has profound implications across several scientific disciplines. This research provides a crucial piece of the puzzle in understanding the remarkable proton conductivity of phosphoric acid, a phenomenon often lauded as "Nature’s proton highway."
Deepening Our Understanding of Biological Charge Transport:
- Molecular Basis of Biological Processes: By pinpointing the precise structure of a key player in proton transfer, scientists gain a more fundamental understanding of how protons move within biological systems. This knowledge is vital for comprehending cellular energy production, signal transduction, and metabolic pathways, all of which rely on efficient charge transport.
- Disease Mechanisms: Disruptions in cellular charge transport have been implicated in various diseases. A clearer understanding of the molecular mechanisms involved could pave the way for novel therapeutic strategies.
Advancing Technological Frontiers:
- Next-Generation Energy Technologies: The exceptional proton conductivity of phosphoric acid makes it a prime candidate for advanced energy applications, particularly in fuel cells. The findings of this study offer a valuable reference point for designing and synthesizing new materials with even greater proton conductivity, potentially leading to more efficient and cost-effective fuel cell technologies.
- Battery Design: Improved understanding of charge transport in phosphate-based materials could also inform the development of next-generation batteries, enhancing their performance, longevity, and safety.
Refining Computational Chemistry:
- Benchmarking Quantum Chemical Models: The experimental data from this study serves as a critical benchmark for validating and improving quantum chemical models. By comparing theoretical predictions with precise experimental results, researchers can refine their computational tools, making them more accurate and predictive for future molecular studies. This iterative process of experimentation and theory is the engine of scientific progress.
A Chronology of Discovery (Inferred):
- Early 2000s – Present: Growing recognition of the critical role of charge transport in biological systems and the potential of phosphate-based materials for technological applications. Theoretical models begin to explore the mechanisms of proton shuttling.
- Mid-2010s: Advancements in experimental techniques, particularly in low-temperature spectroscopy and nanodroplet isolation, become feasible for studying fragile molecular systems.
- Late 2010s – Early 2020s: The collaborative research effort focuses on the H₃PO₄·H₂PO₄⁻ dimer as a key initiating molecule for proton shuttling.
- Recent Period (leading up to publication): Synthesis of the dimer, experimental investigation at near absolute zero temperatures using helium nanodroplets and infrared spectroscopy, coupled with rigorous quantum chemical calculations.
- Publication of Findings: The research team publishes their groundbreaking results, revealing the singular stable structure of the dimer and its implications.
Statements from Related Parties (Inferred and Representative):
While direct quotes from all involved parties are not available in the provided text, one can infer the sentiment and importance of the findings.
A hypothetical statement from a lead researcher might read: "For years, we’ve known that phosphoric acid is a phenomenal conductor of protons, but the precise molecular architecture enabling this efficiency has been somewhat of a black box. By pushing the boundaries of cryogenic experimental techniques and integrating them with cutting-edge computational chemistry, we’ve finally been able to peer into the very heart of this process. The discovery of a single, stable structure for this key dimer challenges some of our prior assumptions and provides a much clearer roadmap for understanding and engineering efficient proton transport."
A representative from a fuel cell industry consortium might comment: "This fundamental research is precisely what drives innovation in the energy sector. Understanding the molecular-level details of proton conductivity in materials like phosphoric acid is invaluable. It provides the scientific bedrock upon which we can develop more robust, efficient, and cost-effective fuel cell membranes and electrolytes, accelerating the transition to a cleaner energy future."
Broader Impact and Future Directions:
The implications of this discovery extend far beyond the immediate scientific community. By demystifying a fundamental aspect of charge transport, this research opens new avenues for innovation in:
- Biotechnology: Understanding and potentially manipulating charge transfer in biological systems could lead to new diagnostic tools and therapeutic interventions for diseases linked to cellular electrical dysfunction.
- Materials Science: The insights gained can guide the design of novel materials with tailored proton conductivity for applications ranging from sensors and catalysts to advanced electronic components.
- Fundamental Physics and Chemistry: The study contributes to a deeper understanding of intermolecular forces, hydrogen bonding dynamics, and the behavior of matter at extremely low temperatures, pushing the boundaries of fundamental scientific knowledge.
The meticulous work of the Fritz Haber Institute and its collaborators has not only illuminated a critical aspect of "Nature’s proton highway" but has also laid the groundwork for future advancements, promising a ripple effect across diverse scientific and technological landscapes. The journey to fully harness the power of charge transport is ongoing, but this recent breakthrough marks a significant stride forward.















Leave a Reply