Plants, the silent powerhouses of our planet, dedicate the majority of their existence to the remarkable process of photosynthesis, converting sunlight into the energy that sustains life. Yet, during the nascent stages of their development, from a mere seed to a fragile seedling, this vital process is not yet operational. In this critical, albeit brief, transitional period, young plants rely entirely on a different energy source: stored fatty acids. The intricate cellular machinery responsible for breaking down these essential fatty acids resides within specialized, membrane-bound compartments known as peroxisomes. Intriguingly, these same cellular organelles are also fundamental components of human cells, hinting at shared biological pathways and potential therapeutic avenues.
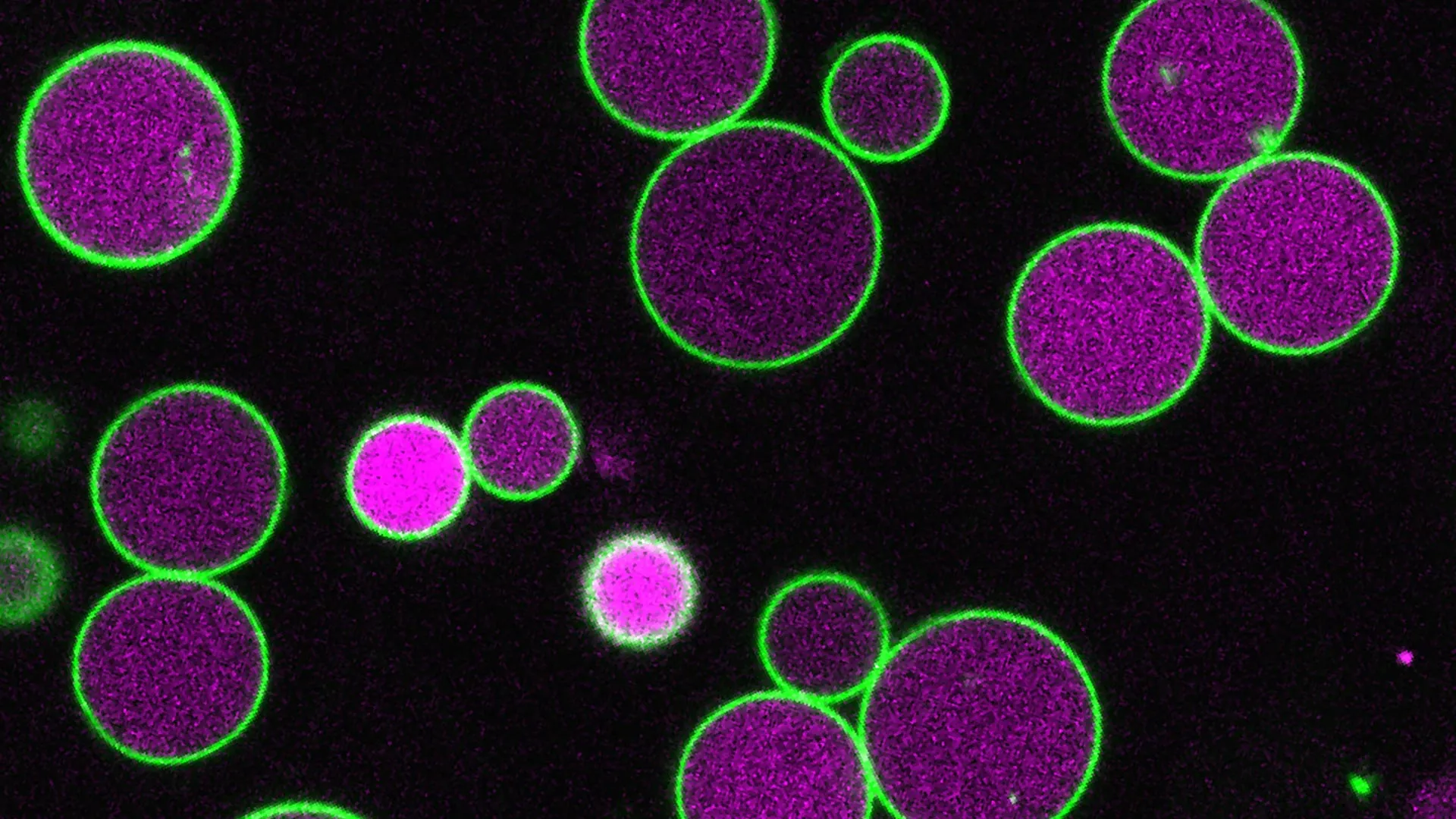
The robust and readily observable nature of plant cells, particularly those of the model organism Arabidopsis thaliana, has long made them an invaluable asset for scientific inquiry into the complex workings of peroxisomes. "The plant we use, Arabidopsis, has large cells and peroxisomes so large that we can see inside them with a light microscope," explained Bonnie Bartel, the Ralph and Dorothy Looney Professor of Biosciences at Rice University. "The peroxisome gets even larger during the seed to seedling stage, when the plant is relying on fatty acids for energy, before shrinking back down to its normal size once the plant can photosynthesize." This dynamic change in peroxisome size, directly correlated with the plant’s metabolic state, presents a unique opportunity to study the mechanisms governing organelle growth and regulation.
The Pivotal Role of Protein PEX11 in Peroxisome Dynamics
At the heart of this research lies the protein PEX11, a key player in the intricate dance of peroxisome life. While scientists had previously established PEX11’s involvement in peroxisome division, the recent groundbreaking work by Bartel’s team has unveiled its equally critical role in controlling the expansion and contraction of these organelles during the crucial seed-to-seedling transition. Their findings, published in the prestigious journal Nature Communications, shed new light on the sophisticated mechanisms that govern organelle size and function.
Nathan Tharp, the lead author of the study and a graduate student in Bartel’s lab, underscored the broader significance of peroxisome research. "Peroxisomes are implicated in some human diseases and used in bioengineering," Tharp stated. "They can, however, be rather tricky to study." This inherent complexity necessitates innovative approaches to dissect their functions.
Navigating the Challenges of Studying a Multi-Gene Protein with CRISPR
Understanding the precise function of a protein often involves a direct experimental approach: deactivating the gene responsible for its production and observing the resulting cellular or organismal phenotypes. However, the case of PEX11 presented a formidable challenge. Unlike many proteins encoded by a single gene, PEX11 in Arabidopsis is produced by a family of five distinct genes. This redundancy meant that the inactivation of just one gene had a negligible impact on the plant’s development or peroxisome behavior. Conversely, attempting to disable all five genes simultaneously proved to be a lethal endeavor, with the plants failing to survive. This critical limitation made it exceedingly difficult to isolate and attribute specific functions to the PEX11 protein.
To overcome this genetic hurdle, Tharp ingeniously employed cutting-edge CRISPR-based gene editing techniques. This advanced technology allowed for the precise and selective disruption of different combinations of the five PEX11 genes. "I was able to use recent advances in CRISPR to go in and break specific combinations of the five genes," Tharp elaborated. "It was only then that we were able to see that PEX11 is clearly involved in controlling the growth of the peroxisome during the seed to seedling stage." This meticulous approach, a testament to scientific ingenuity, finally provided the researchers with the ability to dissect the multifaceted role of PEX11.
Giant Peroxisomes: Unveiling the Mechanism of Growth Control
The experimental manipulation of PEX11 gene combinations yielded striking observations. Tharp successfully engineered two distinct lines of mutant plants, each exhibiting a specific subset of PEX11 genes rendered non-functional. In these mutant plants, the peroxisomes did indeed undergo the expected expansion during the crucial seed-to-seedling growth phase. However, a critical deviation occurred: instead of returning to their normal, smaller size as observed in wild-type plants, these peroxisomes continued to grow, often exceeding normal dimensions dramatically. In some extreme cases, the peroxisomes expanded to such an extent that they stretched across the entire width of the plant cell, transforming into colossal cellular structures.
Furthermore, these abnormally enlarged peroxisomes were characterized by a conspicuous absence of vesicles. These vesicles, small membrane-bound sacs, are typically observed forming within the peroxisome during the breakdown of fatty acids. Their normal presence and formation are believed to be intrinsically linked to the regulation of peroxisome size. Under typical conditions, the budding and formation of these vesicles appear to facilitate the removal of portions of the peroxisome’s outer membrane, thereby contributing to its controlled shrinkage after the period of high metabolic demand.
"The vesicles taking pieces of membrane as they form may help control the peroxisome’s growth," Tharp explained. "In our PEX11 mutants, these vesicles either don’t form or are abnormally small and rare, and so we see these massive peroxisomes, way larger than normal." This crucial observation provided the missing piece of the puzzle, revealing that PEX11, in conjunction with vesicle formation, orchestrates a delicate balance between peroxisome expansion and contraction. The absence of proper vesicle activity, triggered by PEX11 dysfunction, leads to uncontrolled growth, highlighting a fundamental mechanism of organelle size regulation.
A Conserved Mechanism: Implications Beyond Plant Biology
The implications of these findings extend far beyond the realm of plant science. Tharp, driven by scientific curiosity, investigated whether the observed growth control mechanism involving PEX11 and vesicle formation might be conserved across different species. To test this hypothesis, he introduced the yeast homolog of the protein, known as Pex11, into the mutant plant cells that displayed enlarged peroxisomes. The results were profoundly significant.
"We put yeast Pex11 into our mutant plant cells to see if it could return the peroxisomes back to normal," Tharp reported. "And it did." The introduction of yeast Pex11 effectively rescued the mutant phenotype, restoring the peroxisomes to their normal size and function in the plant cells. This remarkable cross-species rescue strongly suggests that Pex11 plays a functionally analogous role in yeast as it does in plants, despite the significant evolutionary divergence between these two kingdoms of life.
This discovery carries substantial weight for understanding human biology and the development of novel bioengineering strategies. "Finding that this protein fills the same role in yeast and plant cells suggests that it may be a highly conserved protein," Bartel emphasized. "Our findings in plants, in this relatively easy-to-study model, may thus be applicable to human cells and cells used for bioengineering." The conservation of this fundamental mechanism implies that PEX11 proteins in other organisms, including humans, likely govern peroxisome size and function through similar pathways.
Broader Impact and Future Directions
The research opens up several exciting avenues for future investigation and application:
-
Human Disease Research: Peroxisomal disorders, such as Zellweger syndrome and X-linked adrenoleukodystrophy, are serious human diseases linked to peroxisome dysfunction. A deeper understanding of peroxisome growth control mechanisms, illuminated by this plant study, could provide crucial insights into the molecular basis of these conditions and potentially lead to the development of new therapeutic targets. For instance, if human PEX11 is found to regulate peroxisome size in a similar manner, therapies aimed at modulating its activity or restoring proper vesicle formation could be explored.
-
Bioengineering Applications: Peroxisomes are increasingly being engineered for various biotechnological applications, including the production of biofuels, pharmaceuticals, and industrial enzymes. The ability to precisely control peroxisome size and dynamics, as demonstrated by this research, could significantly enhance the efficiency and yield of these bioengineering processes. For example, understanding how to promote or inhibit peroxisome expansion could be critical for optimizing their capacity for metabolic product synthesis.
-
Evolutionary Biology: The conservation of the PEX11 functional role across plants and yeast offers a compelling case study in evolutionary biology. Further research could explore the precise molecular differences and similarities in PEX11 function across a wider range of species, providing a deeper understanding of how fundamental cellular processes have been maintained and adapted over evolutionary time.
-
Fundamental Cell Biology: This study contributes significantly to our fundamental understanding of organelle biogenesis and homeostasis. The intricate interplay between protein function, membrane dynamics, and vesicle trafficking in regulating peroxisome size provides a model for studying similar processes in other cellular organelles.
The research by Bartel’s team, rooted in the elegant simplicity of plant biology, has unexpectedly unearthed a fundamental mechanism of cellular regulation with profound implications for human health and advanced biotechnological development. By unraveling the secrets of how peroxisomes grow and shrink, scientists are moving closer to harnessing these vital cellular compartments for the betterment of society. The journey from a tiny seed to a photosynthesizing plant has revealed a sophisticated cellular strategy that resonates across the tree of life, underscoring the interconnectedness of biological systems and the enduring power of scientific discovery.















Leave a Reply